|
Serial dilution involves repeatedly mixing known amounts of source culture with (sterilised) liquid. 1 ml added to 9 ml gives a 10-fold dilution; 1 ml added to 99ml.  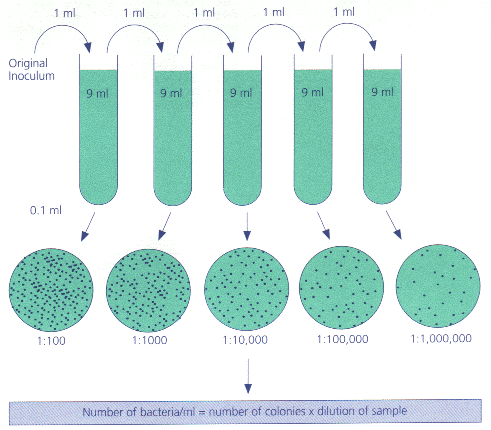
Spectrophotometry & Dilutions Microbiology 542 -- Eric Martz Goals: • Understand how to calculate optimal dilutions. • Determine precision and accuracy of dilutions. • Determine protein concentration with spectrophotometer. • Pros and cons of conventional spectrophotometer vs. 96-well plate absorbance reader. Download the sims 3 pc tasik game minecraft. • Verify instrument performance with simple tests. Optimal Dilutions. Reagent dilutions are needed daily in biochemical or immunological lab work. It is important to know how to design and perform dilutions which are accurate and which meet the needs of the situation. Single Dilution Exercise. You are provided with a concentrated stock solution of a yellow solute (p-nitrophenol), labeled 'NP Stock'. Make a dilution which reduces the concentration by 37-fold, which is a 1/37 dilution. • What do you need to know? Before making your dilution, make a list of things you need to know, and check it with your instructors. (Suppose you are in a research lab and you need to make a dilution of a concentrated reagent stored in the refrigerator. What do you need to know in order to make a dilution which will meet the needs of the situation?) • Record the serial numbers of the pipeters you use (if they have serial numbers); else label the pipeters with tape and marker. (Why is this important?) • Each student (not each pair) should make the 1/37 dilution four times, identically, to test reproducibility. Put 200 microliters of each of your four dilutions, in duplicate, in column 2 (first student) or column 8 (second student) of your 96-well plate (total 8 wells/student) II. Serial Dilutions. • Inspect your 96-well plate. Rinse top and bottom if it is dirty. Be careful not to scratch the bottom with a paper towel -- scratches increase absorbance! Flip rinse water vigorously into the sink (ask an instructor to demonstrate); if done properly, this leaves no significant volume in the wells. • Select one of your four 1/37 dilutions. In the 96-well plate, make three serial dilutions as follows in column 3 (first student) or 9 (second student). Into wells A, B, C put 0.1 ml diluent. Add 0.1 ml of the 1/37 dilution to well A and mix by pipeting in and out. Remove 0.1 ml from well A and add it to well B; mix. Similarly transfer 0.1 ml from B to C, mixing. Remove 0.1 ml from well C and discard it so all four wells contain the same total volume. • Record the serial numbers of the pipeters you use (if they have serial numbers); else label the pipeters with tape and marker. (Why is this important?) • Repeat the serial dilutions three times, so each student now has four replicate series of dilutions (first student in columns 3, 4, 5, 6; second student in columns 9, 10, 11, 12). • Put 0.2 ml of diluent in well A1, which the reader assumes to be the blank. • After you have completed parts I and II, take your plate to one of the 96-well plate readers and read the entire plate twice at wavelength 405 nanometers. Data analysis & comprehension questions. • Does it matter whether you discard 0.1 ml from well C after mixing? After all, the concentration stays the same either way. • Is 405-nanometer light visible? What color is it? Why is 405 nm suitable for measuring absorbance of NP (which is yellow)? • From one reading, determine the mean, SD, and SEM for all 4 of your 1/37 dilutions. Is this mean significantly different from that of your lab partner? • Write the mean, SD, SEM from the previous step on the blackboard. When at least 4 students have written their results on the blackboard, find the highest and lowest. Are they significantly different? If so, what are the possible explanations for the discrepancy? • From one reading, determine the mean, SD, and SEM for the four replicates of the last (row C) of your serial dilutions.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
